
Helaina is raising a $15-25M Series B-1 convertible note financing led by Primary Venture Partners to accelerate its transition from early commercialization to scaled manufacturing and revenue growth. The note includes an 8% annually compounded interest with a 24-month maturity, converting at the next equity financing or at a change of control. Additionally, there’s an added incentive of a $100M post-money valuation cap if the investment amount is $2.5M and above.
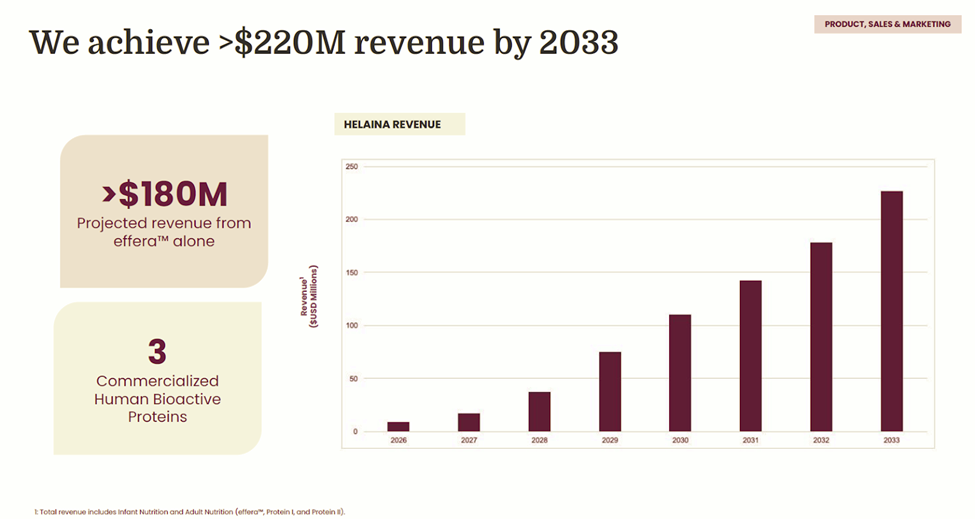
Near-term growth is already accelerating: Helaina forecasts $8.85M in revenue in 2026 and expects to reach EBITDA positivity at $12.5M by 2029. The company projects scaling to $220M in revenue by 2033, with its flagship hero ingredient effera® expected to drive more than 80% of total sales. The raise funds a critical execution window as it expands production capacity, deepens commercial traction, and advances toward large enterprise supply agreements. The company has continued support from existing investors and strategic stakeholders aligned with building next-generation nutrition and longevity infrastructure, alongside new capital focused on scaling precision fermentation and functional health platforms.
A central pillar of the investment is Helaina’s strategic partnership with Nestlé to commercialize human-identical bioactive proteins across infant and medical nutrition, supported by a $9M Joint Development Agreement spanning 2026-2027 to accelerate manufacturing scale and cost reduction. Comparable partnerships within Nestlé’s nutrition division have historically converted into long-term supply agreements, creating a credible pathway for Helaina to enter global infant and clinical nutrition channels by late 2028 or early 2029. Together with declining production costs and expanding commercial adoption, this financing positions Helaina to emerge as a core upstream supplier of bioactive proteins for functional health and longevity-focused nutrition, setting the stage for a meaningful volume inflection beginning in 2030.
INVESTMENT TIMELINE
Virtual Company Presentation
Wednesday, February 18th, 2026
12:00 pm ET // 9:00 am PT
RSVP to Google Calendar invite
Call details available here
Final Investment Commitments Due
Friday, February 27th, 2026
We will take commitments on a rolling basis. To secure your allocation, please submit final commitments here.
Funding & Documents Due
Tuesday, March 17th, 2026
At the end of the commitment period, you will receive details regarding closing documentation and wiring instructions via Carta.
The Company's confidential financing documents and diligence materials are available for review in Carta. Please request access to data room materials at the top of the page. All documents are confidential and not for further distribution. Plum Alley Ventures Company reserves the right to not proceed with the investment opportunity if the $500,000 syndication minimum is not met.
To learn more, watch the recent update presented to Beyond Innovation by Founder & CEO Laura Katz below.
* Engineering Human Biology at Scale: Precision-fermentation platform producing human-identical bioactive proteins, led by effera® (human lactoferrin), with AI-driven strain engineering delivering ~80% productivity gains and ~70% COGS reduction.
* Riding Functional Health & Longevity Tailwinds: Structural shift from commodity protein to clinically backed bioactives across immunity, gut health, women’s health, and longevity optimization is fueling demand for next-gen functional ingredients.
* From Lab to Live Revenue: Commercial inflection underway with $1.4M in first-year sales, 15 active customers, and a 40+ customer pipeline representing $11M near-term revenue potential. Helaina projects $8.85M in revenue in 2026 and expects to reach EBITDA positivity at $12.5M in 2029, marking a clear path from early traction to scalable, profitable growth.
* Massive & Expanding Opportunity: Positioned within an $800B+ global nutrition market and fast-growing functional health segments exceeding $10B, with significant whitespace for human-bioactive proteins.
* Cost Curve Set to Flip the Market: First to scale human-identical lactoferrin with costs trending toward and below bovine supply, enabling superior efficacy at commodity pricing and potential category dominance.
* Strategic Partner with Global Reach: Deep Nestlé partnership includes $9M in non-dilutive funding and pathway to a potential $10-50M annual supply agreement, reinforcing enterprise demand and validation.
* Team DNA Built to Scale Beyond R&D: Domain expert founder-led with a strengthened executive bench spanning PepsiCo, IFF, AB InBev/ZX Ventures, and leading nutrition ingredient operators.
Helaina is building a category-defining platform for the production of human-identical bioactive proteins, designed to unlock a new generation of functional ingredients for global nutrition, health, and longevity markets. Founded in 2019, the company uses precision fermentation and proprietary strain engineering to manufacture proteins structurally equivalent to those found in human biology, beginning with lactoferrin, a multifunctional protein linked to immune modulation, iron regulation, and microbiome health. By engineering biology rather than extracting from animal sources, Helaina addresses longstanding supply constraints and cost barriers that have historically limited the widespread use of clinically relevant bioactive proteins.

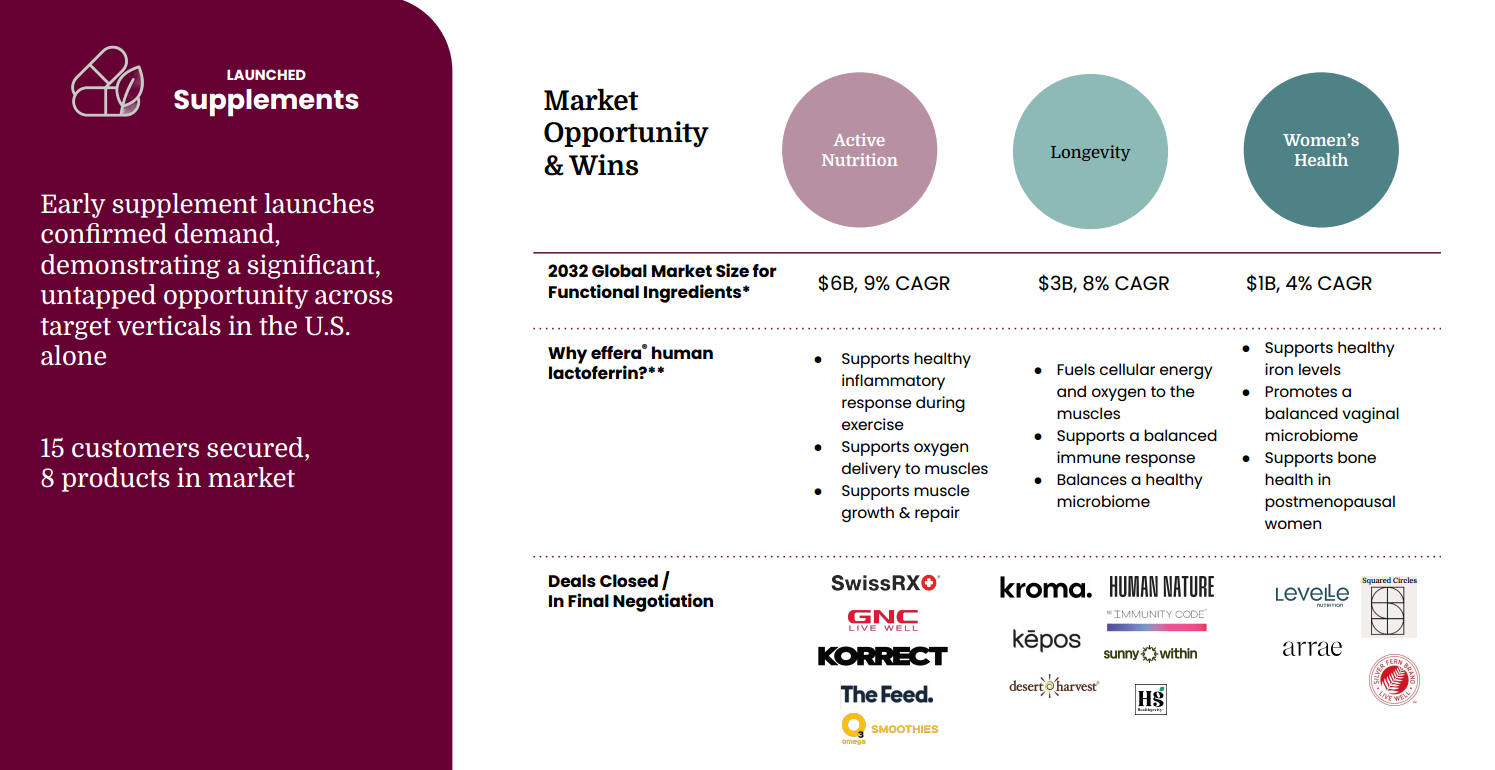
The company has now crossed a key inflection point from R&D-stage biotech to early commercial scale. In 2025, Helaina generated $1.4M in revenue with eight products in market across 15 active customers in the adult nutrition segment, validating both demand and pricing power for high-function bioactive ingredients. Early launches across women’s health, active nutrition, and longevity categories demonstrate strong commercial pull from premium supplement and wellness brands seeking differentiated, science-backed ingredients capable of supporting immune function, iron metabolism, gut health, and inflammatory balance.
To date, Helaina has commercialized three human bioactive proteins, including effera® (human lactoferrin), osteopontin, and alpha-lactalbumin. effera®, the company’s lead ingredient, has already secured placement across multiple supplement SKUs and continues to expand into new formats and applications, with additional product categories under development spanning functional foods, beverages, and personal care. This multi-protein roadmap positions Helaina to evolve from a single-ingredient company into a broader platform supplier for next-generation functional nutrition.
Helaina has also secured strategic partnerships across manufacturing, distribution, and enterprise nutrition that provide both technical validation and long-term commercial leverage. Most notably, the company has established a joint development relationship with Nestlé, expanded through $9M in non-dilutive R&D funding, with $2M recognized in 2025 and an additional $7M expected across 2026 and 2027. This collaboration accelerates production scale, supports cost reduction initiatives, and creates a credible pathway into global infant and medical nutrition channels. The partnership also positions Helaina to be among the first to introduce human-identical lactoferrin into next-generation infant formula, with broader expansion potential into adult nutrition and therapeutic nutrition applications over time.
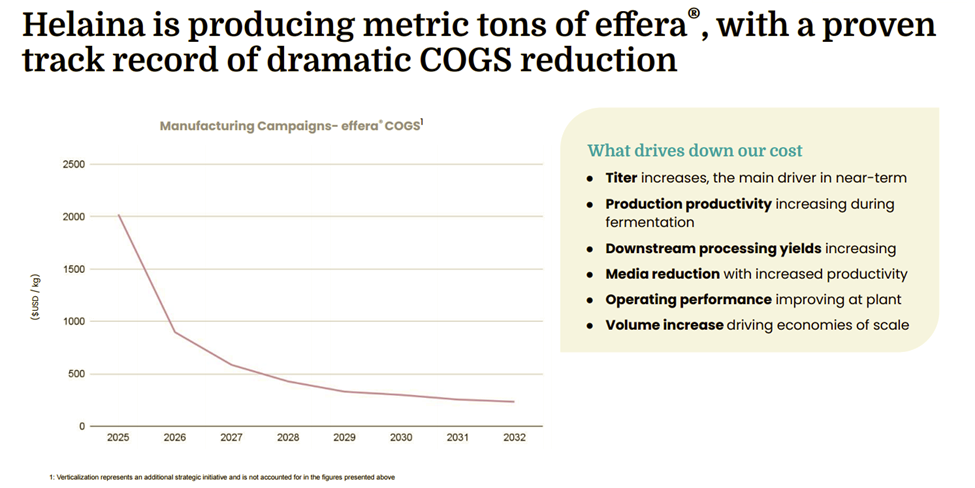
Operationally, Helaina has demonstrated significant progress in scale and unit economics, transitioning to commercial-scale precision fermentation while driving major improvements in productivity, titer, and yield, and achieving roughly a 70% reduction in manufacturing costs. These advances, combined with growing commercial momentum and strategic partnerships, position Helaina as an emerging revenue-generating platform with a clear path toward scaled supply, global distribution, and multi-protein commercialization, establishing its bioactive protein platform as core infrastructure for next-generation functional nutrition and longevity-focused health products.

The market opportunity for bioactive protein manufacturing sits at the intersection of functional nutrition, preventative health, and the rapidly expanding longevity economy. Global nutrition is undergoing a structural shift away from commoditized macronutrients toward clinically supported, functional ingredients that deliver measurable outcomes such as immune resilience, gut integrity, metabolic health, and recovery. Consumers and health systems alike are prioritizing preventative interventions that extend healthspan, creating demand for bioactive compounds that move beyond basic nutrition and into performance-oriented health optimization. Within this context, bioactive proteins represent a new category of high-value inputs capable of reshaping the composition of supplements, functional foods, and medical nutrition. Like probiotics, collagen, and creatine before them, human bioactive proteins are poised to define a new functional ingredient category, with the potential to follow a similar trajectory from niche innovation to mass-market adoption and sustained, high-growth expansion.
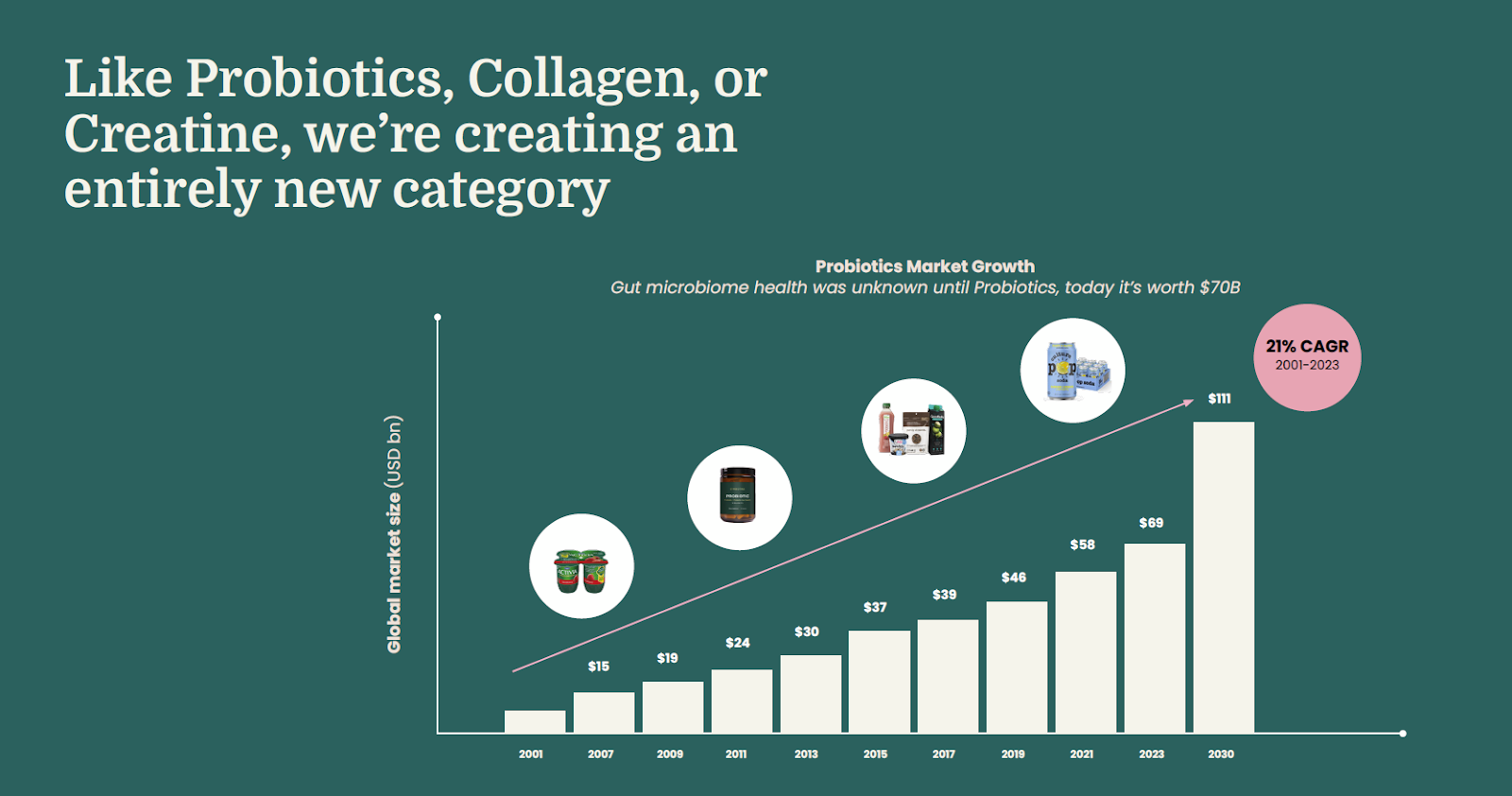
Near-term demand is concentrated across four high-growth verticals, including active nutrition, gut and immune health, and women’s health, each experiencing strong consumer adoption and sustained premium pricing dynamics. These categories provide an attractive entry point for next-generation proteins that can command higher price points while delivering differentiated functional benefits. At the same time, enterprise demand from global food and nutrition companies is increasing as incumbents seek more reliable, scalable, and ethically sourced alternatives to animal-derived specialty proteins that are often supply constrained and price volatile.

The longer-term opportunity extends well beyond today’s premium supplement niche. As precision fermentation lowers production costs and clinical validation expands, human bioactive proteins have the potential to scale into mass-market functional foods, infant and maternal nutrition, and medical nutrition. This mirrors the trajectory of earlier functional ingredients that began as premium inputs and ultimately became foundational across everyday consumer health products. The ability to produce human-identical proteins at scale could significantly expand total addressable market by enabling broader use across everyday nutrition and preventative health products.

Functional ingredients such as probiotics, collagen, and omega-3s demonstrate the scale of what a single breakthrough ingredient can become. Together, they have helped build a global functional food and supplement market exceeding $500B, with individual categories now worth tens to hundreds of billions annually. Probiotics alone are projected to surpass $130B globally by 2032, while collagen and omega-3 markets are expected to respectively reach $16B+ and $6B+.
Over time, the category is poised to become core infrastructure for longevity-focused nutrition and functional health. As consumer demand shifts toward products that support long-term health optimization rather than short-term supplementation, clinically validated bioactive ingredients are expected to see accelerating adoption. Companies capable of manufacturing these proteins reliably and at scale stand to capture significant share across a converging market spanning consumer wellness, clinical nutrition, and next-generation functional food systems.
Helaina Platform
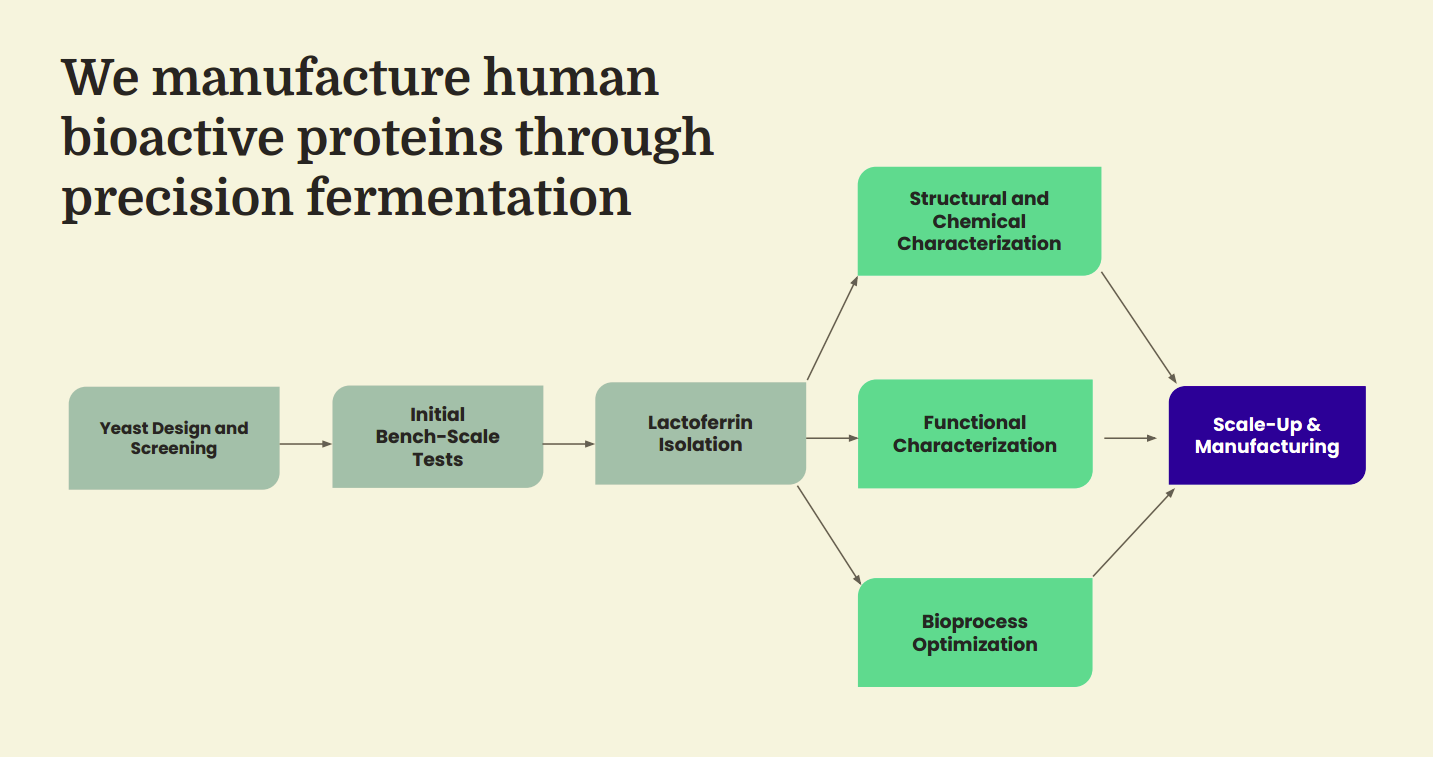
The Helaina platform is a vertically integrated precision fermentation and glycoengineering system designed to produce complex human bioactive proteins at industrial scale. At its foundation is a proprietary glycoengineered yeast host that has been programmed to synthesize human-identical proteins with the correct amino acid sequence, structural folding, and glycosylation patterns required for biological activity. By recreating these proteins through controlled microbial fermentation rather than animal-derived extraction, the platform enables reliable supply, consistent quality, and a structurally faithful product that mirrors native human biology.
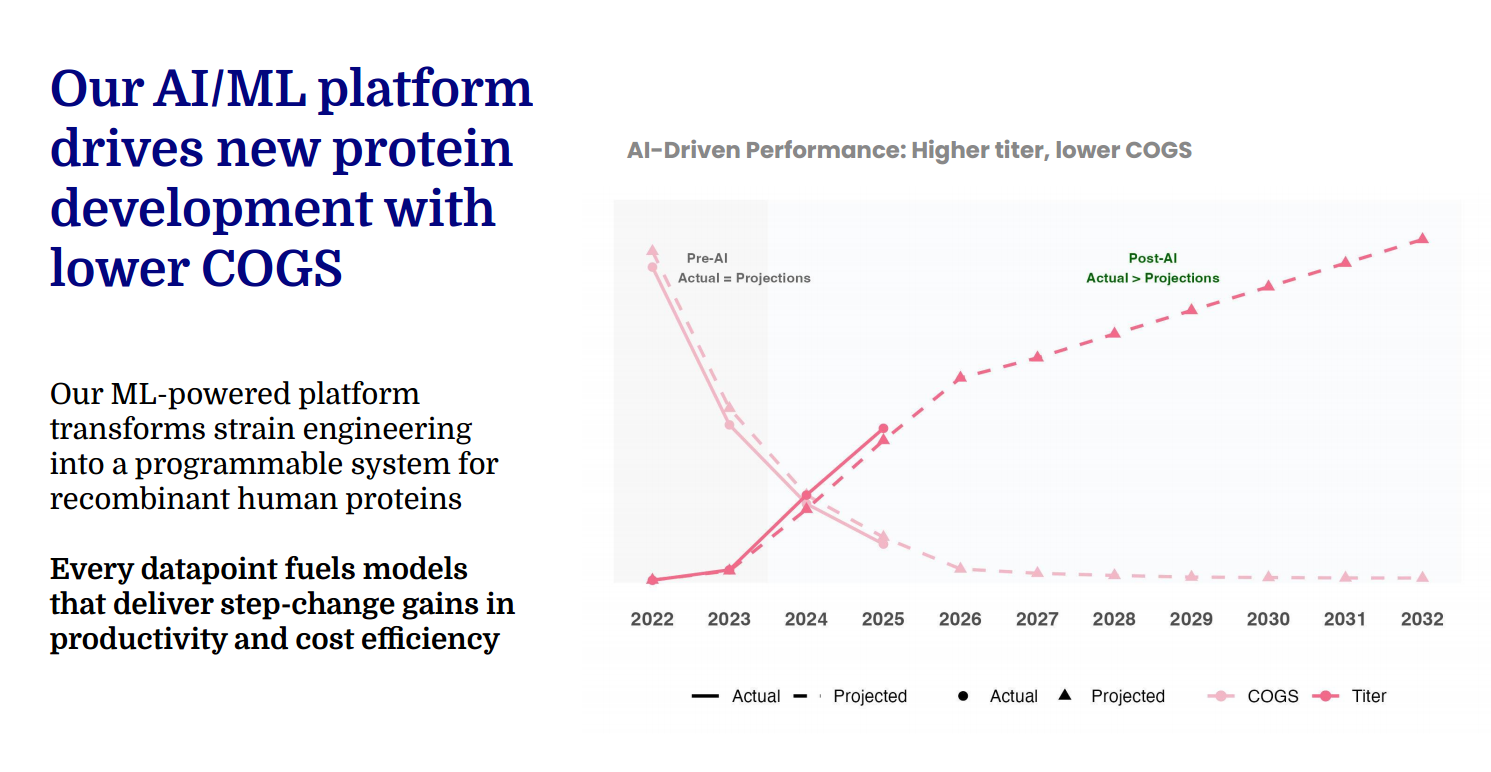
This biological engine is reinforced by a purpose-built data and machine learning infrastructure that captures and structures experimental and manufacturing data across the entire development lifecycle. Predictive models trained on proprietary multi-omics and bioprocess datasets guide strain design, optimize fermentation conditions, and forecast scale-up performance. The result is a closed-loop system in which engineered biology, data infrastructure, and manufacturing continuously inform one another, enabling rapid iteration, cost reduction, and performance improvement. As the dataset expands and models become more precise, the platform compounds its advantage, accelerating development of future proteins while strengthening process efficiency and reliability.
Machine Learning & Data Infrastructure
A proprietary data and machine learning infrastructure underpins the entire development and manufacturing engine, transforming complex biological production into a predictive, closed-loop system. Structured datasets spanning genomics, transcriptomics, metabolomics, and bioprocess parameters are continuously captured and integrated into unified databases that serve as a single source of truth across R&D and manufacturing. Machine learning models trained on this high-fidelity dataset analyze multidimensional biological and process variables to identify non-obvious bottlenecks, prioritize high-performing genetic configurations, and forecast production outcomes. The result is a data-driven architecture that shortens development timelines, reduces experimental burden, and materially increases the probability of achieving optimal production performance.
These predictive capabilities extend directly into commercial-scale biomanufacturing. Once optimized strains are selected, machine learning-guided simulations and scale-down models inform fermentation strategies across bioreactor environments, optimizing parameters such as oxygen transfer, feed rates, and temperature to maximize protein expression and consistency. Digital-twin process models allow production scenarios to be tested virtually before implementation, enabling efficient transfer from laboratory to commercial scale while minimizing operational risk and variability. Downstream purification workflows then isolate and refine the target protein through centrifugation, filtration, and chromatography, with analytical validation confirming sequence accuracy, glycan positioning, and structural conformation to ensure fidelity to the intended human biological profile.
Beyond cost and scalability advantages, this integrated platform enables measurable functional improvements over traditional animal-derived proteins, including improved bioavailability, reduced immunogenic response, and greater stability through digestion. Together, engineered biology, machine learning, and scalable manufacturing form a compounding system in which improved strains generate richer datasets, and richer datasets continuously refine predictive models and process performance. This closed-loop architecture enables reliable, cost-efficient production of complex human bioactive proteins with high fidelity, consistent quality, and industrial scalability.
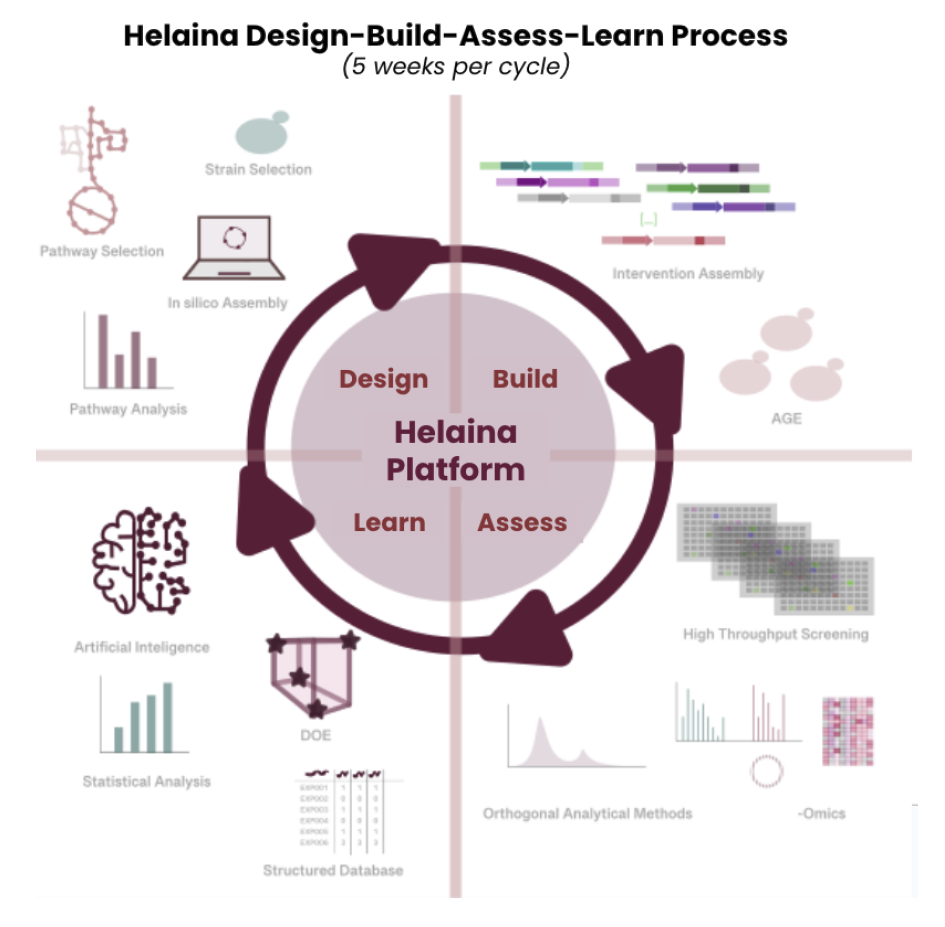
R&D Process: From Strain Design to Commercial Manufacturing
Helaina’s R&D engine is built around a closed-loop Design-Build-Assess-Learn (DBAL) framework that integrates advanced genome engineering, machine learning, and commercial-scale process development. In the design phase, computational models and multi-omics datasets identify high-impact genetic targets and metabolic pathways. During build, modular DNA assembly and CRISPR-enabled editing rapidly generate engineered strains across a wide design space. The assess stage leverages high-throughput screening and analytical validation to evaluate yield, glycosylation, and functional performance across thousands of variants. Insights from each cycle feed directly into the learn phase, where structured datasets refine predictive models and guide subsequent engineering iterations. This DBAL system enables continuous gains in titer, productivity, and strain stability while reducing development timelines and experimental burden.
Once leading strains are identified, the process transitions seamlessly into bioprocess development and manufacturing optimization. Machine learning-guided simulations and scale-down models determine optimal fermentation parameters across bioreactor environments, enabling efficient transfer from laboratory to commercial scale. Operational excellence in manufacturing is achieved through tightly controlled upstream fermentation and downstream purification workflows that prioritize consistency, yield, and cost efficiency. Continuous monitoring, data feedback loops, and process standardization ensure that production improvements are sustained and replicated across manufacturing campaigns.

Together, the DBAL-driven R&D engine and disciplined manufacturing operations form an integrated development and production system. Early-stage biological innovation feeds directly into scalable manufacturing, while late-stage, real-world production data continuously informs the next generation of strain design and process optimization. This closed-loop architecture enables rapid iteration, sustained cost reduction, and reliable industrial-scale production of complex human bioactive proteins with high fidelity and operational rigor.
Overview
Helaina is building the first scalable platform for human bioactive proteins, using precision fermentation to recreate the functional proteins found in human colostrum and throughout the body. To date, the company has commercialized three human-identical proteins, effera® (lactoferrin), osteopontin, and alpha-lactalbumin, forming the foundation of a next-generation nutrition stack designed to align with human biology rather than animal-derived or synthetic substitutes. Together, these ingredients enable more precise support for iron metabolism, immune function, gut integrity, and early-life development across both infant and adult nutrition.
Helaina has moved beyond R&D into commercialization. Helaina now has eight products on market spanning infant formula and adult nutrition, demonstrating early validation across multiple regulatory pathways and customer segments. These launches position Helaina among the first companies to deliver human-equivalent bioactive proteins at commercial scale for functional foods, supplements, and specialized nutrition, establishing credibility as both a technology platform and ingredient supplier

effera® serves as the first commercial proof point for a broader pipeline of human-equivalent functional proteins. Helaina is assembling a modular portfolio that spans infant formula, adult nutrition, and longevity-focused products, with additional proteins in development and fermentation efficiency steadily improving. As costs decline and production scales, the company is positioning itself as a foundational supplier of next-generation functional ingredients across global health and nutrition markets.
Over time, Helaina’s platform extends well beyond supplements into functional foods, medical nutrition, and personal care. By enabling scalable access to human-identical bioactive proteins, the company is shifting nutrition from reactive supplementation toward proactive biological optimization, with the potential to establish bioactive proteins as core inputs for immune resilience, metabolic health, and long-horizon longevity.
effera™ Human Lactoferrin
Helaina’s flagship product, effera®, is a human-identical lactoferrin produced through precision fermentation and designed to deliver the functional benefits of one of the most biologically important proteins in human nutrition. Lactoferrin plays a central role in iron regulation, immune modulation, inflammation control, and gut barrier integrity, yet traditional supply has been constrained by the cost and inefficiency of extraction from bovine milk. Helaina’s platform enables scalable production with superior purity, consistency, and functional performance, unlocking entirely new commercial and clinical applications while making this historically scarce bioactive accessible at meaningful scale.

effera® is positioned as a foundational ingredient across active nutrition, women’s health, gut and immune health, and longevity-focused formulations, with initial commercialization centered on the U.S. dietary supplement market. In active nutrition, it addresses iron deficiency, inflammation, and recovery challenges common among high-performance and female athletes, supporting improved energy, endurance, and recovery. In women’s health, effera® is being deployed across PMS, prenatal, menopause, and hormonal health formulations where inflammation-driven iron dysregulation affects mood, metabolic function, and performance, opening whitespace for products that move beyond symptom management toward root-cause biological optimization.
Within longevity and preventive health, effera® introduces a differentiated mechanism centered on inflammatory pathway modulation, iron optimization, and immune resilience. Early positioning highlights its potential role in dampening chronic inflammation and supporting muscle, bone, metabolic, and immune function over time. As a highly bioavailable, human-identical protein suited for daily, long-duration use, effera® aligns with the rapidly expanding longevity and biohacking category, where consumers increasingly prioritize foundational inputs that support systemic health rather than single-endpoint outcomes.
Clinical Validation & Safety
effera™ is supported by a growing body of preclinical and human clinical research confirming structural equivalence, safety, and functional efficacy.
Key validation highlights include:

A recent double-blind clinical study confirmed that effera™ does not trigger anti-human lactoferrin antibodies, even at doses significantly above recommended intake, enabling safe continuous daily use without tolerance or cycling. Additional safety reviews and expert panel recommendations support its use as a conventional food and supplement ingredient with no evidence of toxicity or adverse outcomes.
Strategic Positioning
Helaina operates a vertically integrated B2B ingredient platform built on precision-fermented, human-identical bioactive proteins. Rather than selling finished consumer products, the company supplies high-function proteins as core inputs to leading nutrition, supplement, and functional food brands. This upstream model allows Helaina to concentrate on protein development, manufacturing, and process optimization while leveraging partners’ established distribution, regulatory infrastructure, and brand equity. Revenue is generated through ingredient sales and long-term supply agreements, creating recurring demand as partners integrate Helaina’s proteins across multiple SKUs and product lines.
As production efficiency improves and clinical data expand, Helaina expects to move down the cost curve into higher-volume applications across functional foods, medical nutrition, and infant formula, expanding both addressable market and operating leverage. Its commercial strategy follows a land-and-expand model, typically beginning with a single product integration or pilot launch and scaling into multi-SKU adoption and larger supply agreements as partners gain confidence in performance, demand, and economics. This dynamic positions Helaina as a core upstream supplier rather than a specialty ingredient vendor, with each successful launch increasing switching costs and embedding its proteins deeper into partner portfolios, while enabling the company to commercialize additional bioactive proteins through the same manufacturing and partner network to build a multi-protein platform with cumulative revenue streams and shared infrastructure.
Commercial Validation
Helaina has reached a clear commercial inflection point, transitioning from an R&D-stage biotech into a revenue-generating ingredient platform with accelerating market adoption. In its first full year of sales, the company generated approximately $1.4M in revenue across 15 active customers and multiple products in market, signaling strong willingness to pay for human-identical bioactive proteins even at premium pricing. A pipeline of more than 40 prospective customers with meaningful annual revenue potential provides forward visibility as additional SKUs launch and existing partners scale, particularly within high-growth functional health categories where Helaina’s ingredients are positioned as core differentiators rather than commodity inputs. The company projects $220M in revenue by 2033, with flagship ingredient effera® expected to account for more than 80% of total sales.
effera™ has been designed for seamless integration into functional foods, beverages, and supplements. Its 98% purity, neutral taste and odor, and low inclusion rates enable incorporation across capsules, powders, beverages, gummies, bars, and performance nutrition formats without reformulation challenges. Precision fermentation has also unlocked a scalable manufacturing pathway for human lactoferrin, historically one of the most supply-constrained bioactive proteins. As production volume scales, yield improvements and manufacturing optimization are driving a steady decline in unit costs. Combined with low inclusion rates and high potency per dose, this supports improving gross margins for downstream brands and expanding accessibility over time.
As the cost curve continues to decline, effera™ is positioned to transition from a premium niche ingredient to a broadly adopted foundational input across the global functional nutrition and longevity markets, with precision-fermented human bioactives representing a new category of scalable, biology-aligned ingredients.
Partnerships
Commercial momentum continues to build through a deepening pipeline of prospective customers and expanding integrations with early partners. Initial adopters across wellness and performance nutrition are incorporating Helaina’s human lactoferrin into flagship formulations, validating efficacy-driven demand and sustained pricing power. As manufacturing costs decline and clinical data strengthens structure-function positioning, growth is expected to accelerate through customer expansion, new product launches, and broader adoption across premium and mid-tier price points.
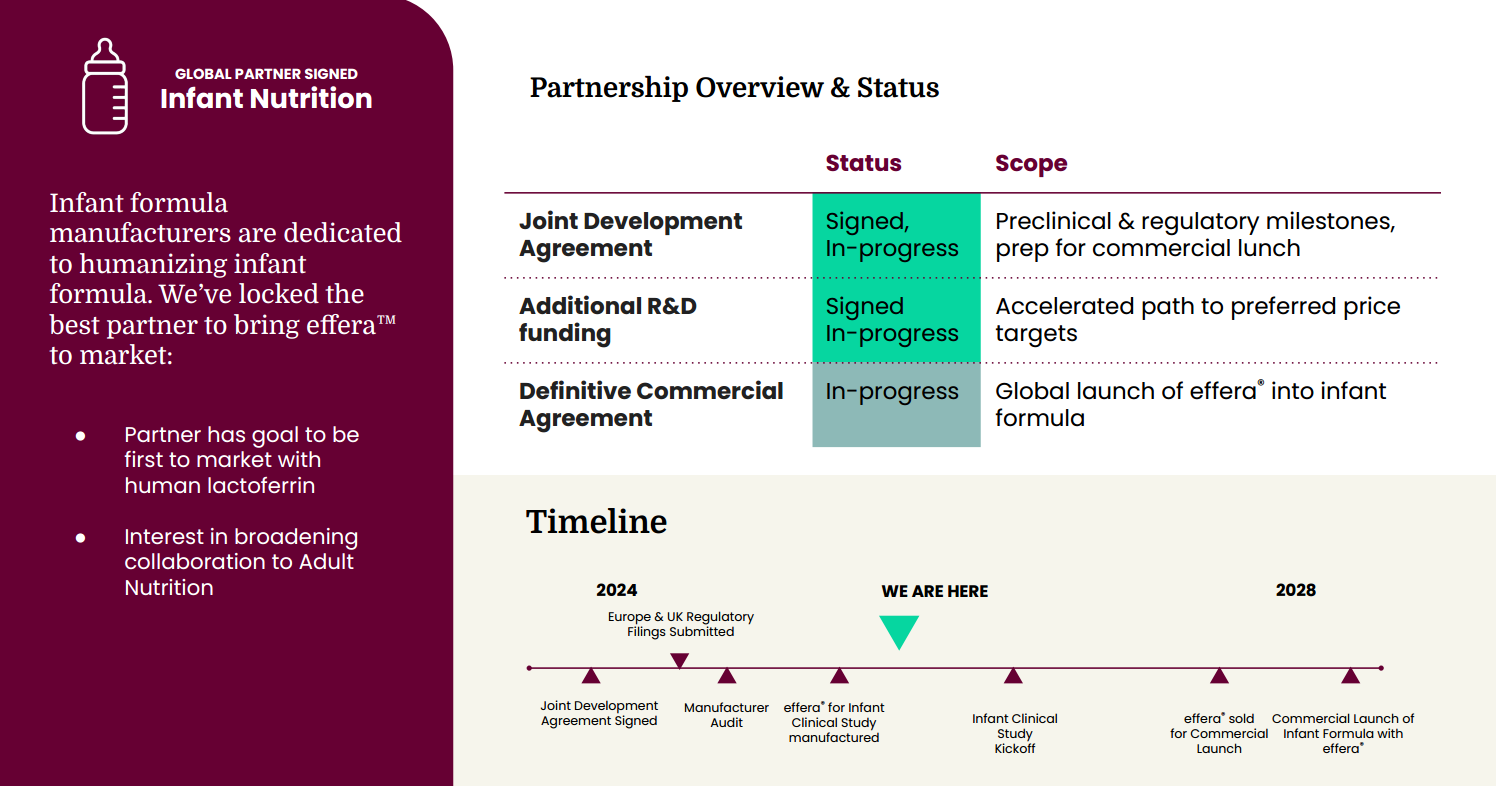
Strategic partnerships underpin Helaina’s long-term positioning. Collaborations with consumer health brands are seeding demand, while fermentation and manufacturing alliances support scalable production. At the center of this ecosystem is the company’s joint development partnership with Nestlé. Historically, comparable programs within Nestlé’s nutrition division have progressed into multi-year supply agreements once technical and regulatory milestones are achieved, positioning Helaina as a potential core upstream supplier across global nutrition categories.
Underpinning this trajectory is a defensible foundation of biomanufacturing and protein engineering IP spanning strain design, glycosylation control, and precision fermentation processes required to produce human-identical bioactive proteins at scale. Combined with the operating leverage of an ingredient supplier model, this IP moat positions Helaina to transition from early market entry to scaled, repeatable supply and to emerge as a category-defining provider of bioactive proteins for functional health and longevity-oriented nutrition.
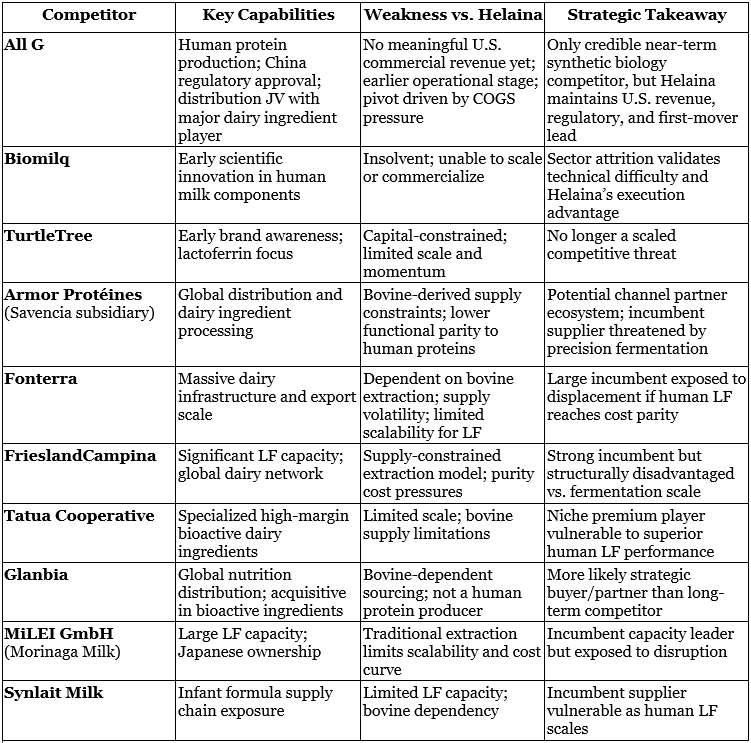
The competitive landscape for bioactive protein manufacturing has rapidly consolidated as technical complexity, capital intensity, and scale-up challenges have acted as a sector-wide filter. Over the past 12-18 months, several high-profile entrants pursuing human-identical or precision-fermented proteins have exited or retrenched, underscoring the difficulty of producing complex human proteins at commercial scale. Today, All G stands as the only direct, credible competitor developing human-identical bioactive proteins for nutrition and functional health; however, Helaina maintains a meaningful first-mover advantage, with US commercial revenue, active customer adoption, and established manufacturing and regulatory pathways already in place, while many peers remain earlier in commercialization or have pivoted toward lower-cost verticals such as cosmetics.
Beyond All G, the remaining competitive set is primarily composed of adjacent players across bovine-derived lactoferrin, specialty dairy ingredients, and global nutrition supply chains, as outlined in the table. These include legacy producers and distributors with strong infrastructure and customer relationships but structural constraints tied to animal-based supply, including scalability limits, cost volatility, and purification complexity. As precision fermentation reaches cost parity and clinical differentiation strengthens, human-identical proteins are positioned to displace traditional animal-derived inputs across high-value nutrition categories.
Helaina is raising a $15-25M Series B-1 convertible note financing led by Primary Venture Partners, positioning the company at the inflection point between early commercial validation and scaled ingredient production. The note includes an 8% annually compounded interest with a 24-month maturity, converting at the next equity financing or at a change of control. Additionally, there’s an added incentive of a $100M post-money valuation cap if the investment amount is $2.5M and above.
In 2021, Plum Alley first invested in Helaina’s $22.17M Series A at a $66.17M post-money valuation. To date, the company has raised $83M, achieved FDA GRAS status in 2024, and advanced toward commercial-scale production through strategic partnerships, including a $9M Joint Development Agreement with Nestlé. Primary Venture Partners has led every round since inception, with participation from tier-one crossover and growth investors including Spark Capital, known for backing category-defining technology platforms, and Avidity Partners, a leading life sciences and biotech public-equity investor. This continued institutional support reflects conviction in Helaina’s ability to build a foundational bioactive protein platform for global nutrition and functional health markets.
The current financing is structured to drive near-term, high-confidence execution across manufacturing scale, clinical validation, and enterprise customer expansion. Core priorities include reducing cost of goods below $1,000/kg through AI-enabled strain and process optimization, completing clinical studies to support differentiated structure-function claims, and scaling commercial adoption across active nutrition, practitioner channels, and global strategic partners. These initiatives are expected to materially improve gross margins, accelerate customer conversion, and position Helaina for enterprise-scale supply agreements.

Laura Katz, Founder & CEO: Laura is a food scientist and category creator who identified the “human-equivalent protein” gap years before the broader functional nutrition and bioactive protein markets began to mature. NYU’s youngest-ever adjunct professor in Food Science, she combines deep technical credibility with consumer product development expertise from roles at NUGGS, Plated, and Dylan’s Candy Bar. Since founding Helaina in 2019, Laura has led the company from early platform development through commercial launch of its flagship human lactoferrin ingredient, effera®, positioning Helaina at the forefront of precision-fermented bioactive proteins and building the strategic partnerships required to scale across global nutrition markets.
Anthony Clark, PhD, CTO: Anthony is the architect of Helaina’s manufacturing and fermentation engine, bringing more than two decades of industrial biotechnology and R&D leadership across PepsiCo, IFF, Firmenich, and MycoTechnology, where he served as CSO. He has a track record of scaling novel fermentation technologies from lab to commercial production and now leads Helaina’s process optimization, cost reduction, and industrial-scale manufacturing strategy. At Helaina, Anthony is responsible for driving the company’s path to cost parity and eventual cost leadership versus traditional animal-derived proteins.
Taylor Burroughs, CFO: Taylor brings rigorous financial and operational discipline from leadership roles as CFO of NotCo and VP of Finance at ZX Ventures (AB InBev), an organization known for its data-driven operating model and capital efficiency. Since joining Helaina in 2025, he has strengthened financial planning, burn management, and strategic capital formation while supporting commercial scaling and long-term manufacturing strategy. Taylor leads finance, RevOps, and capital strategy, including future facility financing and vertical integration initiatives critical to Helaina’s next phase of growth.
Dan DeMarino, VP of Commercial: Dan is a seasoned commercial operator in the supplement and functional ingredient ecosystem, with more than a decade of experience spanning Deerland Probiotics, TSI Group, and American River Nutrition. He has played a central role in translating Helaina’s complex scientific value proposition into commercial adoption, leveraging deep industry relationships to secure early enterprise customers and build a high-velocity pipeline across active nutrition, women’s health, and practitioner channels. His credibility and network within the industry have been instrumental in accelerating revenue growth and positioning effera® as a category-defining ingredient.
Claire Carlson, Head of Strategy: Claire is a strategic operator with a background in management consulting and cross-functional leadership at Helaina, where she has served in multiple roles including Chief of Staff to the CEO. She leads corporate strategy, enterprise partnerships, and high-impact initiatives, including negotiations with global partners such as Nestlé. Known for her operational range and execution discipline, Claire plays a key role in aligning Helaina’s scientific, commercial, and strategic priorities as the company scales into a global bioactive protein platform.
DISCLAIMER: This material is provided solely for informational and educational purposes to support independent investment research and evaluation. It does not constitute, and should not be construed as, investment advice, a recommendation, an offer to sell, or a solicitation to buy any security or investment product. No fiduciary relationship is created by the provision of this information. Recipients are responsible for conducting their own diligence and for making independent investment decisions in consultation with their legal, tax, financial, and other professional advisors. Any views, projections, or forward-looking statements are subject to change without notice and are inherently uncertain. Information presented may be derived from third-party sources believed to be reliable but has not been independently verified, and no representation or warranty is made as to its accuracy or completeness. Investing involves risk, including potential loss of principal. By accessing this material, you acknowledge that it is for research purposes only and agree not to rely on it as the primary basis for any investment decision.